WHAT HEALTH PROFESSIONALS NEED TO KNOW:
Highlights from the OECD/EUIPO Report on Trade in Counterfeit Pharmaceutical Products
John B. Hertig, PharmD MS, CPPS, FASHP
Associate Professor and Vice-Chair of Pharmacy Practice, Butler University College of Pharmacy and Health Sciences
Shannon L. Castek, PharmD
PGY2 Pharmacy Resident, Ambulatory Care and Education, RL Roudebush VAMC and Butler University College of Pharmacy and Health Science
HOW PERVASIVE IS THE PROBLEM?
According to the United States Centers for Disease Control and Prevention, nearly 74% of office visits result in providing, prescribing or continuing a medication. In these visits, approximately 2.9 billion medications, or pharmaceuticals, were prescribed in the U.S. annually (CDC, National Ambulatory Medical Care 2016 National Summary Tables ). The supply chain supporting this significant volume is complex and increasingly global. Unfortunately, criminal enterprises have long recognized the lucrative potential inherent in this business.
Global trade in counterfeit pharmaceuticals was valued at 4.4 billion USD in 2016. According to the Organization for Economic Co-operation and Development (OECD) and the European Union Intellectual Property Office (EUIPO), “illicit trade in fake goods is a growing threat in a globalised and innovation-driven economy, and poses major health and safety threats to consumers.” The OECD and EUIPO conducted analytical studies designed to foster better understanding of the challenges in securing the supply, trade and use of pharmaceuticals. Their findings were published in the 2020 “Trade in Counterfeit Pharmaceutical Products” report*, covering the scale of the problem, factors contributing to a rise in counterfeit pharmaceuticals and efforts to combat the problem. For the purposes of that report, “counterfeit pharmaceuticals” are defined as products deliberately and fraudulently mislabeled in identity and/or source. The vast majority of counterfeit medications contain either active ingredients in inappropriate concentrations or undisclosed active ingredients, posing serious threats to public health. This summary will highlight key aspects of the report, with a focus on health professionals.
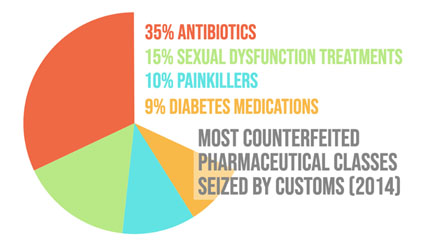
PRIMARY TARGETS: MEDS AND MARKETS
Data from the report illustrates counterfeit antibiotics, lifestyle drugs (those related to the treatment of conditions such as baldness, wrinkles, erectile dysfunction, or acne) and painkillers are most targeted by counterfeiters. Treatments for malaria, diabetes, epilepsy, cardiovascular diseases, allergies and cancer are also widely counterfeited. Preliminary research conducted by these authors suggest medications on shortage and those that have limited/restricted access are prime targets. Categories of pharmaceuticals targeted by counterfeiters continues to expand, including more expensive “specialty” therapies. It is widely assumed that high-income countries can effectively limit these products from their markets due to strong regulatory frameworks. A World Health Organization (WHO) analysis contradicts this assumption, however, as cases of counterfeit incidents revealed upper and lower-middle income countries comprised a vast majority (93%) of all cases.
Counterfeiters succeed by exploiting weaknesses in fragmented supply chains. The OECD/EUIPO report notes the rapidly expanding use of mail and courier (small-parcel) services to transport counterfeit pharmaceuticals worldwide. Consumer-driven e-commerce and pervasive internet access has furthered the distribution of pharmaceutical products (both legitimate and counterfeit) online. Bolstered by the ability to remain anonymous behind the veil of a web address on the freely accessible surface web or covert “dark web,” counterfeiters gain easy access into markets. Meanwhile, consumers consistently demonstrate a willingness to take risks buying products online, disregarding consequences of purchasing products from unknown sources. Online purchases have appeal for a variety of reasons, including speed and convenience, potential cost savings, avoiding discussing sensitive conditions and frequent absence of a prescription requirement. There are an estimated 35,000-40,000 active online pharmacies, with 600 added every month. U.S. studies assert a significant increase (greater than four-fold) in the number of people buying medicines online. Illegal online sales have moved from websites to social media platforms. From 2015-2018, more than 10,000 Facebook accounts selling counterfeit Pfizer medications were identified, while during a six-month period in 2018, 1,000 Instagram accounts were also reported.

AVOIDING PATIENT HARM: A FOCUS ON ILLEGAL ONLINE PHARMACIES
The OECD/EUIPO report notes illegal online (or rogue) pharmacies present a unique and increasing challenge, with Europe and the United States appearing to be main targets. Health professionals should be aware of these trends, recognizing illegal online pharmacies threaten patient safety. The WHO estimates over half of medicines purchased over the internet from illegal sites are counterfeit. These medications can affect individuals in a variety of ways, including adverse reactions from improper ingredients, development of antimicrobial resistance and progression of infections. Counterfeit products may also lead to therapeutic failure, thereby increasing morbidity, mortality and prevalence of disease. Estimates from the report show that between 72,000 and 169,000 children die from pneumonia every year after receiving counterfeit medication. Fake anti-malarial medication is likely responsible for 116,000 deaths worldwide. Additional consequences include a loss of confidence in health systems, increased health system spending to overcome outcomes of counterfeit medications, decreased productivity and lost income due to prolonged illness or death. Today, fear associated with the COVID-19 pandemic has led more consumers online for treatments, cures, tests and protective equipment.
A 2017 survey of U.S. consumers revealed that only a small fraction (5%), were familiar with resources aimed to identify safe online pharmacies. Given this small proportion of patients, paired with the growing number of consumers looking to buy pharmaceuticals online, healthcare professionals are encouraged to always ask patients where they obtain their medications. This query can proactively identify at-risk patients and open the conversation to facilitate education regarding legitimate avenues for obtaining pharmaceuticals. Further, resources are available to help patients stay safe online. The National Association of Boards of Pharmacy has acquired the top-level domain name “.pharmacy.” Patients need only “look to the right of the dot” to find “.pharmacy” and be confident in the online seller. Other organizations such as the Alliance for Safe Online Pharmacies Global and Legit Script have resources and tools to inform health professionals and keep patients safe when purchasing medication online.
The pharmaceutical industry is extremely prone to counterfeiting. Criminals utilize the internet as a guise, while consumers are unaware of the risks and potential for harm associated with the trade of counterfeit pharmaceuticals online. A lack of deterrent penalties, the expansion of e-commerce and misuse of small parcels in trade all contribute to this challenge. The OECD/EUIPO report helps thought-leaders make informed decisions in the face of the ever-changing and rapidly expanding global trade in counterfeit pharmaceuticals. All stakeholders, including healthcare professionals and consumers, must recognize the dangers associated with deviating from a safe source of medication.
*References throughout to the OECD/European Union Intellectual Property Office, Trade in Counterfeit Pharmaceutical Products, 2020 unless otherwise stated.
THE BRAND PROTECTION PROFESSIONAL | JUNE 2020 | VOLUME 5 NUMBER 2
COPYRIGHT 2020 MICHIGAN STATE UNIVERSITY BOARD OF TRUSTEES